The FDA and its peculiarities in the cleaning and steam sterilization processes recommended in the IFU
Medical products that must be cleaned and steam sterilized (autoclave) prior to use or in the case of thermolabile products, cleaned and disinfected prior use, are those marketed as non-sterile or those products marketed as sterile but subject of reprocessing. For products in these conditions, it is mandatory that the instructions for use contain the step-by-step for cleaning and the details and parameters for proper sterilization or disinfection. The suitability of the instructions is the manufacturer responsibility, who must guarantee a sterile product at the time of use through the validations of each method.
If your company intends to enter the North American market, you should know about some very specific points required by the FDA regarding validation processes and consequent recommendations in the instructions for use. To this end, the FDA has the guide titled: “Reprocessing Medical Devices in Health Care Settings: Validation Methods and Labeling, March 17, 2015”.
In general, the FDA understands that for the data from these validations to be reliable, the accessories used in those validations must have been previously cleared by the FDA (if applicable) and available for purchasing by prospective consumers in the United States. What accessories are we talking about? From the detergent to be indicated for cleaning, disinfectant for high level disinfection, to the wrapping or surgical grade paper used during sterilization, biological indicators, chemical indicators, among others.
Specifically for sterilization, the FDA has compiled possible parameters to be validated based on the accessories already registered in the United States, ie, steam sterilization validations should be made at a very specific temperature and sterilization time as well as drying time post-sterilization, depending on autoclave technology, whether gravity displacement or dynamic air removal. See below:
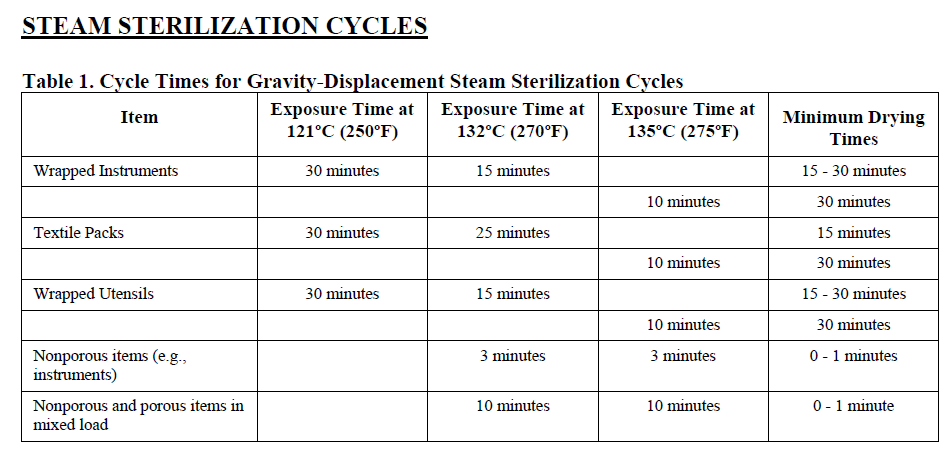

Complicated?
Talk to us. Passarini Regulatory Affairs can help you.
The FDA guide on this topic is available at the link below:
https://www.fda.gov/media/80265/download
For more information and explanations about it, send us an e-mail: contato@passarini.com.br
Passarini Regulatory Affairs
Published: 08/29/2019
You may also be interested in:


