Service Regulation in Uruguay
Manufacturers of medical devices that wish to distribute and market their products in Uruguay must comply with Uruguayan legislation, that is, for a medical device to enter the country, it must obtain the health registration issued by the country’s health authority (Ministry of Public Health – M.S.P.).
Passarini Group, through its technical team in Uruguay, offers the comprehensive advice and support you need to comply with the regulatory framework in all its stages.
Discover some of the services we provide in Uruguay:
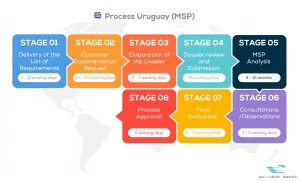
See below our complete MSP process flow
Passarini Group is a company that has the best specialists in Regulatory Consultancy of Medical Products, applying the One Stop Shop concept daily in your company with practical solutions.


